Sensa Platform
Build Wearable Medical Devices Faster
A ready-to-deploy wearable platform combining hardware, firmware, SDK, and cloud.
How Sensa works
One integrated platform. Hardware to insights.
Sensa is a full-stack wearable platform built entirely in-house by ITR. Three core components work together to accelerate development while maintaining regulatory compliance from day one.
Device Platform
Multiple hardware platforms optimized for different wearable use cases: suitable for a wide range of on-body device needs, including SensaRing for 24/7 multi-sensor wear, SensaPatch for clinical ECG and respiratory monitoring, SensaBand for wrist-worn continuous monitoring, SensaTip for episodic monitoring, and SensaNeuro for EEG applications.
SensaSDK
Cross-platform (iOS, Android, Windows) software development kit for rapid app development. Handles Bluetooth connectivity, local data buffering, real-time streaming, and automatic cloud synchronization. No RF expertise required.
SensaHub
Secure cloud platform for data ingestion, storage, visualization, and export. HIPAA-compliant, audit-logged, encrypted at rest. REST APIs enable custom integrations and analytics. Portal for study management and real-time biomarker review.
Why Sensa
Built for MedTech development from day one.
Instead of starting from scratch, developers can build on Sensa's ready-to-deploy wearable platforms.
Developed under medical device development practices (IEC 62304–aligned processes), Sensa enables faster POC/MVP development, early biosignal validation, and the reuse of existing documentation when evolving toward a production-ready device.
Best For
Accelerated Time-to-Market
Reduce development time by ~6 months with proven hardware modules and pre-built cloud infrastructure.
Regulatory-Aligned Development
Development follows IEC 62304 practices; documentation supports your regulatory submissions.
Multi-Disciplinary Integration
Hardware, firmware, mobile, and cloud components designed to work together seamlessly.
Flexible Engagement
Use Sensa for POC validation, integrate into your pipeline, or evolve it into your final product.
Device Platform
The right device for every application.
Choose the hardware platform that best matches your clinical application. Each module combines optimized sensors, wireless connectivity, and power management.
SensaRing
Ultralight, discrete ring form factor maximizes 24/7 wearer compliance. Multi-sensor suite supports algorithm diversity from wellness to clinical research.
- Continuous heart rate and wellness monitoring
- Sleep quality tracking with motion + PPG
- Stress/mental health monitoring via EDA
- Activity and fitness applications
- Research studies requiring multi-modal biosignals

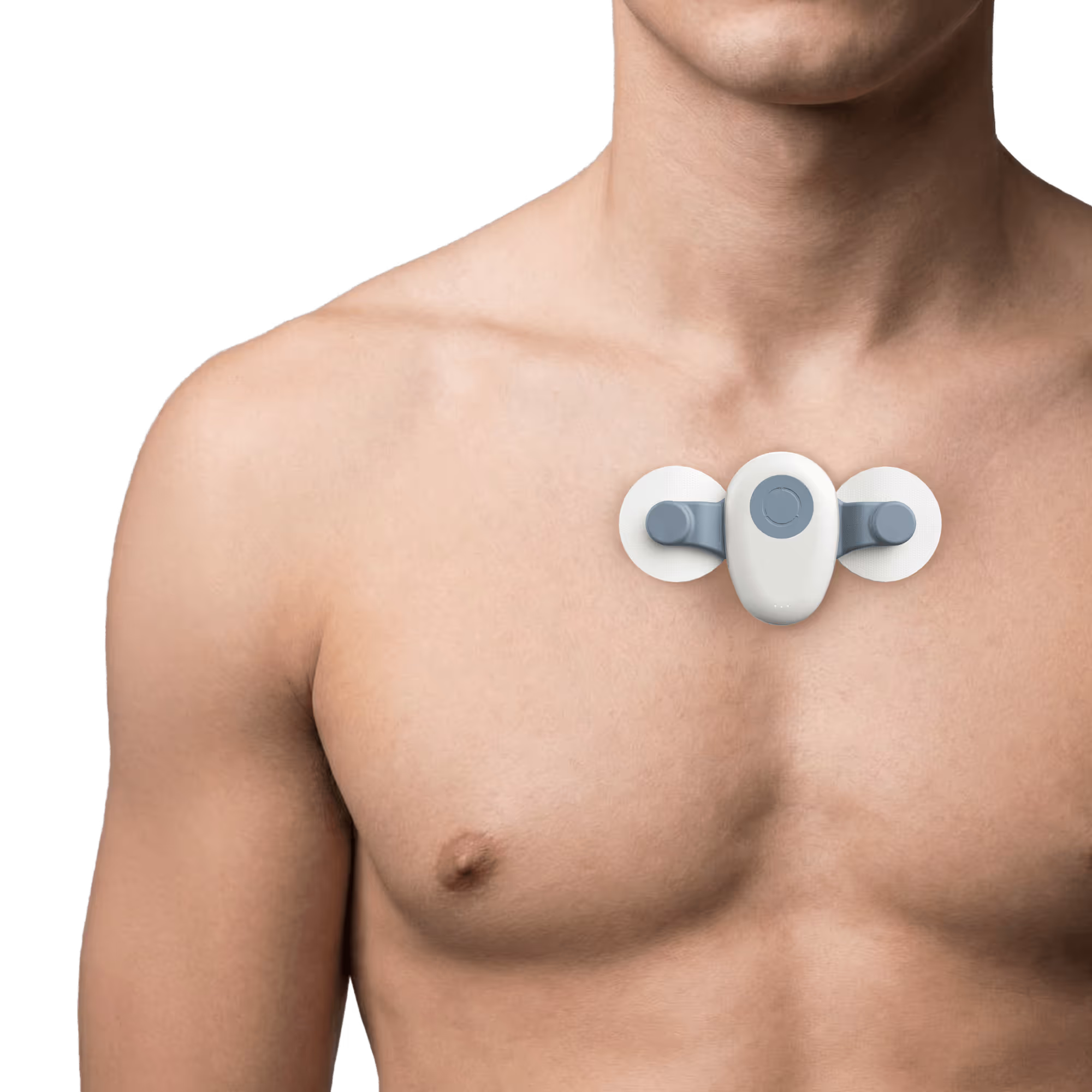
SensaPatch
Chest placement optimal for ECG signal quality. Bioimpedance (BioZ) adds respiratory rate and fluid monitoring. LTE variant enables direct cellular upload without smartphone.
- Holter and Mobile Cardiac Telemetry (MCT)
- Clinical-grade ECG waveform collection
- Remote patient monitoring (RPM) with cellular connectivity
- Home sleep apnea testing (HSAT) workflow support
- Long-term cardiac research
- Continuous respiratory rate monitoring
- Heart failure monitoring
- Thoracic fluid status trending

SensaBand
Familiar wristband form factor maximizes daily adoption. LTE connectivity enables automatic data sync without smartphone dependency.
- All-day heart rate monitoring
- Activity and movement tracking
- Sleep quality assessment
- Remote patient monitoring with built-in LTE
- Continuous wellness applications
- Research requiring activity and physiological context

SensaTip
Ultraportable and discreet. High optical signal quality from direct finger contact. Ideal for research deployments and travel.
- Episodic monitoring (user activates during specific times)
- Research requiring high-fidelity PPG data
- Sleep apnea screening workflows
- Clinical research and feasibility studies

SensaNeuro
Clinical-grade EEG signal quality. Multi-channel recording enables source localization. Research-grade accuracy with minimal setup time.
- EEG-based sleep staging and architecture analysis
- Seizure prediction and detection research
- Cognitive research and attention monitoring
- Mental health monitoring (depression, anxiety markers)
- Brain-computer interface (BCI) research
- Neuroscience clinical trials and studies

From POC to Market
The Sensa-to-Product Development Journey
Sensa is a full-stack wearable platform built entirely in-house by ITR. Three core components work together to accelerate development while maintaining regulatory compliance from day one.
01. POC Development
Market & algorithm validation
WEEKS 1 - 4
What the Founder Does
- Select appropriate Sensa hardware module(s)
- Develop application on SensaSDK
- Deploy to beta users/patients
- Collect real sensor data via SensaHub
ITR Involvement
Advisory support on hardware selection and SDK usage
02. Funding & Validation
Prototype refinement
Months 1 - 3
What the Founder Does
- Raise seed/Series A funding using POC proof
- Conduct small pilot studies or user research
- Validate clinical assumptions with real data
- Finalize product requirements and regulatory pathway
ITR Involvement
None (founder bootstraps with Sensa)
03. Product Finalization
Production-ready engineering
Months 3 - 9
What ITR Does
- Hardware Customization: Form factor refinement, component optimization, PCB layout for manufacturability.
- Firmware Development: Power optimization, algorithm integration, OTA updates, offline caching.
- Compliance & Testing: IEC 60601 safety, EMC testing, biocompatibility, software validation.
- Regulatory Documentation: IEC 62304 design history file, ISO 14971 risk management, FDA/MDR submissions.
- Manufacturing Readiness: DFM analysis, supplier qualification, pilot builds, CMO handoff.
04. Market Launch
Regulatory & manufacturing
Months 9 - 12+
What happens
- FDA 510(k) or CE mark clearance (timing varies by pathway)
- Contract manufacturing ramps production
- Sales and distribution launch
- Post-market surveillance and updates
ITR Involvement
Ongoing support (optional software updates, technical questions, liability documentation)
Autoplay / Paused
Discovery Call · 30 Minutes
Ready to Accelerate Your MedTech Program?
Share your product vision, clinical goals, and timeline. Our team will map a path to market using Sensa and ITR's full-service engineering, compliance, and manufacturing support.